Synthesis of aryl-fused bicyclo[3.1.1]heptanes and validation as naphthyl bioisosteres
Aidan Kerckhoffs, Maud Tregear, Pol Hernández-Lladó, Massimiliano Runfola, Holly Shearsmith, Nils Frank, Marc Panosetti, Sarah E. Squire, Lee Moir, Kirsten E. Christensen, Fernanda Duarte, Kay E. Davies & Angela J. Russell
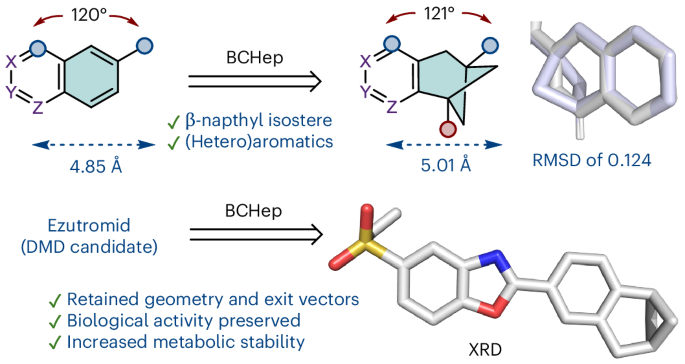
Although naphthalene motifs are frequently encountered in drugs and lead compounds, their flat, sp2-rich nature and susceptibility to cytochrome P450-mediated metabolism often limit their developability. Here we report the study of derivatizable aryl-fused bicyclo[3.1.1]heptanes (BCHeps) as sp3-rich bioisosteric replacements for naphthalene and other fused bicyclic (hetero)aromatics, including underrepresented β-naphthyl units. The BCHeps were efficiently accessed via an intramolecular crossed [2+2] photocycloaddition enabled by visible light energy transfer and subsequently diversified to provide a range of different scaffolds. Here we show that the incorporation of BCHep-based naphthyl isosteres into the AhR antagonist ezutromid preserves key geometric exit vectors while reducing the fraction of sp2 carbon atoms. Importantly, these analogues retain biological activity and display improved metabolic stability towards CYP1A-mediated metabolism. Solid-state structures, cellular assays and microsomal studies confirm that BCHep substitution mitigates reactive metabolite formation, validating aryl-fused BCHeps as true bioisosteric replacements for meta-substituted arenes and 2-naphthalenes.
https://www.nature.com/articles/s41557-026-02129-2